The 10 ps TOF-PET challenge
a step toward reconstruction-less TOF-PET
- a spur on the development of fast timing across disciplines and technologies
- an opportunity to collaborate as a community on a complex final frontier problem
- an incentive and opportunity to raise further funding
- a way to shed light on advanced nuclear instrumentation for medical imaging and beyond
A step towards reconstruction-less TOF-PET
News & Publications
Roadmap toward the 10 ps time-of-flight PET challenge
IEEE TRPMS 2020 Best Paper Award
Overview
Since the seventies, positron emission tomography (PET) has become an invaluable medical molecular imaging modality, especially for cancer diagnosis and the monitoring of its response to therapy and more recently in direct combination with X-ray computed tomography (CT) or magnetic resonance (MR).

PET is an incredibly sensitive imaging technique based on electron-positron annihilations and the nearly coincident detection of the corresponding pairs of photons simultaneously emitted in opposite directions and electronically collimated along a common line, aka. a line of response (LOR). Accumulated detections of different annihilation events along various LORs produce projections of the radioactive concentration volume distribution of a radiopharmaceutical labelled with a positron emitter radioisotope, such as 18F or 11C. Tomographic image reconstruction from all the measured projections then leads to the 3D volume representation of the estimated radionuclide distribution, whose accuracy depends mainly on the ability to associate coincident detections with actual annihilation photons.
The localization of the emission point of an annihilation pair along a LOR depends on the detection of the time difference between the two annihilation photons, also known as the time-of-flight (TOF) difference of the photons, whose accuracy is given by the coincidence time resolution (CTR). This additional information makes the tomographic inversion problem much less ill-posed, hence resulting, hence resulting in a better signal-to-noise ratio of the activity measurement. Taking into account the speed of light, which is approximately 30 cm/ns, a CTR better than 10 picoseconds FWHM will ultimately allow to obtain a direct 3D volume representation of the estimated activity distribution of a positron emitting radiopharmaceutical, at the mm level and without the need for tomographic inversion, thus introducing a quantum leap in PET imaging and quantification.
Moreover, a 10 ps capability will increase effective PET sensitivity, as compared to the state-of-the-art, at least a factor of 16, with the following expected consequences:
- Reduction of the radiation doses of molecular imaging procedures to negligibly low levels;
- Reduction of the synthesized quantity of radiopharmaceutical needed for each examination, and thus of the relatively high cost currently associated with in-vivo molecular imaging procedures;
- Further extension of the benefit of molecular imaging procedures beyond oncology and towards cardiovascular, neurological, metabolic, inflammatory, infectious or metabolic disease (such as diabetes), including in the paediatric, neonatal, and prenatal contexts;
- Maximizing the spatial and temporal resolution of PET based molecular imaging;
- Precise dynamic studies of molecular processes of high interest in pharmacology for screening and selecting candidate molecules for the next generation of drugs or new applications thereof;
- Potentially further extension of molecular in-vivo imaging to study "systems biology" of the whole human body through whole-body imaging systems;
- No need for full angular coverage of the patient, opening many new opportunities for PET system design
While crossing the 10 ps frontier will increase the performance of TOF-PET and lead to the advent of reconstruction-less PET, it will also permit efficient positron range monitoring in hadrontherapy (including with protons), improve the performance of Compton camera developed for conventional scintigraphy and nuclear waste management, and impact both the development of positron annihilation lifetime spectroscopy (PALS) used in material science and the development of laser imaging detection and ranging (LIDAR) systems for automotive solutions, e.g. for self-driving cars. Such a feat will not only require to make progresses on the development of new scintillators (e.g. to produce enough photons with a minimum time jitter in the first 10 ps after their excitation), but also on markedly improving light collection, photo-detection and photo-detector electronic readout.
A number of emerging technologies in the domain of scintillating materials, nanotechnologies, solid state photodetectors and electronics support the idea that crossing the 10 ps frontier is not just a wild dream and that, as hinted at in preliminary studies 1 an ambitious and coordinated effort could, lead to a new generation of TOF-PET scanners with unprecedented performance.

Non-TOF back-projection
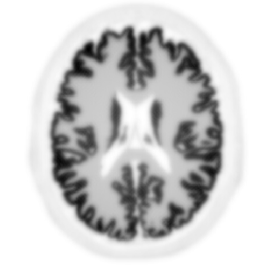
TOF back-projection with 10ps FWHM CTR
Hoffman brain phantom simulated analytically without noise (courtesy: Johan Nuyts, University of Leuven).
Therefore, we intended to launch a challenge to foster the next quantum leap in PET imaging and make reconstruction-less, high-sensitivity PET scanners a reality, thus paving the way towards reducing by an order of magnitude the radiation dose (currently 5-7 mSv for a total-body PET), the scan duration (currently > 10 min), and the scanning cost per patient (currently > 1000 € per scan). Combining 10 ps TOF-PET with other modalities (X-ray CT, MRI, ultrasound, optical) will further enable multi-parametric data acquisition and lead to a paradigm shift for in vivo molecular imaging. This will in turn durably impact societal healthcare challenges not only in oncological disease, but also in neurodegenerative, inflammatory, infectious or metabolic diseases, from prenatal to old age.
Since it was proposed to organize the 10 ps challenge, with the endorsement of numerous colleagues and learned societies, we have seen that this proposal has created a real enthusiasm for research towards this goal, which in itself constitutes an incentive success. But despite the interest of several learned societies, and our repeated efforts, current conditions have not made it possible to secure funding for the challenge and its hosting by a learned society. However, should a non-governmental organization or a learned society wish to rekindle interest in such a challenge, they are invited to contact us.
Endorsements
Companies
Scientists
- Pedro Almeida, University of Lisbon, Portugal
- Etiennette Auffray, CERN, Geneva, Switzerland
- Luc Bidaut, University of Lincoln, UK
- David Brasse, IPHC, CNRS/IN2P3, Strasbourg, France
- Simon Cherry, UC Davis, USA
- Fabrizio Davi, Polytechnic University of the Marches, Ancona, Italy
- Christophe Dujardin, University Claude Bernard, Lyon
- Réjean Fontaine, University of Sherbrooke, Canada
- Sanjiv Sam Gambhir, Stanford University School of Medicine, USA
- Antonio J. Gonzalez, Polytechnic University of Valencia, Spain
- Stefan Gundacker, CERN, Geneva, Switzerland
- Hossein Jadvar, University of Southern California, Los Angeles, USA
- Jae Sung Lee, Seoul National University, South Korea
- Terry Jones, UK
- Nikolaos Karakatsanis, Cornell University, New York, USA
- Mark Ladd, DKFZ, Heidelberg, Germany
- Roger Lecomte, University of Sherbrooke, Canada
- Paul Lecoq, CERN, Geneva, Switzerland
- Homer A. Macapinlac, UT MD Anderson Cancer Center, Houston, USA
- Steven Meikle, University of Sydney, Australia
- Laurent Ménard, IMNC, University Paris Descartes, France
- Christian Morel, CPPM, Aix-Marseille University, Marseille, France
- Johan Nuyts, University of Leuven, Belgium
- Sergey Omelkov, University of Tartu, Estonia
- Jörg Peter, DKFZ, Heidelberg, Germany
- Jean-François Pratte, University of Sherbrooke, Canada
- John Prior, CHUV, University of Lausanne, Switzerland
- Magdalena Rafecas, University of Lübeck, Germany
- Dennis Schaart, Delft University of Technology, Netherlands
- Volkmar Schulz, RWTH-Aachen University, Germany
- Markus Schwaiger, Technical University of Munich, Germany
- Viatcheslav Sharyy, CEA-IRFU, Saclay, France
- Kuangyu Shi, University of Bern, Switzerland
- Gintautas Tamulaitis, Vilnius University, Lithuania
- Marc-André Tétrault, Mass General Hospital, Boston, USA
- Kris Thielemans, UCL, London, UK
- Harry Tsoumpas, University of Leeds, UK
- Stefaan Vandenberghe, University of Gent, Belgium
- Joao Varela, University of Lisbon, Portugal
- Dimitris Visvikis, LaTIM, University of Western Brittany, Brest, France
- Dominique Yvon, CEA-IRFU, Saclay, France
- Karl Ziemons, FH-Aachen University of Applied Sciences, Juelich, Germany












